As part of a new series, I’ll be spending some time trying to review the local “stem cell” clinic offerings to help you figure out high from low-quality offerings. My first installment is a Red Tail wellness flyer sent to me by a local physical therapist. We have treated him and his family so he wanted me to check out this local chiropractic clinic that was offering “stem cells”. Why do I put “stem cells” in quotes? Read on.
The Purpose of this Review
My goal here isn’t to pick on Red Tail Wellness Centers or any specific clinic, but merely to inform consumers. In this case, Red Tail is a pretty typical functional medicine clinic run by a chiropractor that employs a nurse to perform blind injections using an umbilical cord tissue product. There are many other local clinics that fit into this same template, so Red Tail isn’t unique. Let’s dig in.
Who is Red Tail Wellness?
Red Tail is a chiropractic clinic located in Boulder, Colorado where Ian and Karen Hollaman practice. Interestingly, as of today’s date, there is no nurse practitioner nor physician listed on the website. The clinic website says it offers functional medicine, stem cell therapy, chiropractic care, blood analysis, applied kinesiology, and neurofeedback.
What Does Red Tail Say It Does with Stem Cells?
This is what Red Tail’s brochure states:
“Where do you get your stem cells from?
Our Stem cells are derived from two different sources, the Wharton’s Jelly layer of the umbilical cord as well as placental tissue. These cells are sourced from donated birthing tissues from full-term deliveries. Comprehensive medical and social histories of the donor are obtained and tissues are procured, processed, and tested to exceed the standards established by FDA.”
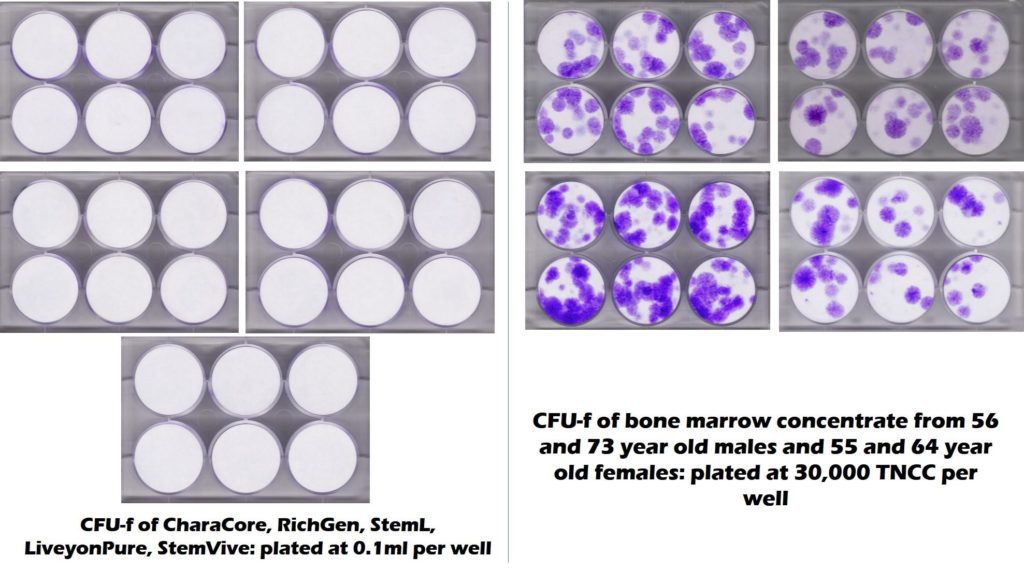
Hence Red Tail claims that it’s using a commercially available umbilical cor/placental product that contains mesenchymal stem cells. However, what do we know about what’s in FDA 361 registered umbilical cord and placental tissue products? Three studies have looked at the stem cell content of birth tissue products and failed to find any live and functional stem cells (Cornell, UC Davis, and our lab) (1-3). In addition, this recently commissioned study performed by the Colorado State University Translational Medicine Institute tested 5 commonly used umbilical cord products that all claimed to have stem cells, but found none (in this picture white=no stem cells).
What was tested that does have stem cells (purple dots) on the right? That’s middle-aged and elderly bone marrow. Hence, it’s very unlikely that Red Tail is injecting anyone with viable mesenchymal stem cells.
The Red Tail Scientific References
The brochure I reviewed also provided two scientific references (4,5). The first is a tiny study of 4 knee arthritis patients treated with isolated and culture-expanded placental mesenchymal stem cells. Regrettably, it’s unknown why Red Tail would use this reference as it has nothing to do with the products that the clinic uses, which are nucleated cell extracts from Wharton’s Jelly. Meaning if the study is about the effects of oranges, Red Tail uses apples. There is zero published evidence that the product (Corecyte made by Predictive Biotech based on the phone call we made) has any viable mesenchymal stem cells or has any clinical data showing it helps knee arthritis patients. The next study is a comparison of isolated and culture-expanded stem cells from bone marrow, adipose, and umbilical cord blood. This is a lab study using rat lung tissue. Regrettably, this study also has nothing to do with the product that Red Tail uses and that has little to do with treating knees, hips, or shoulders.
The Red Tail Quality Matrix
Red Tail charges $4,950 for a knee injection and ancillary rehabilitation and other services. So let’s break down what you get for that money:
- A simple ultrasound-guided knee injection.
- No physician specialist, but a nurse practitioner. I should note that organizations that train doctors in how to perform these orthopedic regenerative medicine organizations like IOF refuse to train NPs due to a lack of background knowledge.
- No stem cells being injected. See above.
- No data that the product it’s using helps any orthopedic condition.
In summary, Red Tail claims to be injecting stem cells but isn’t injecting a product that’s likely to contain any stem cells. Does the product they’re using help arthritis, tendons, or ligaments? Nobody knows at this point as there is no research. From a quality matrix standpoint, instead of getting a specialist physician performing these procedures using x-ray and ultrasound guidance, you’re getting an injection from a nurse.
Related:
Regenerative Medicine of the Rockies Review
Denver Regenerative Medicine Review
Keystone Health & Wellness Review
____________________________________________________________________
References:
(1) Berger D, Lyons N, Steinmetz, N. In Vitro Evaluation of Injectable, Placental Tissue-Derived Products for Interventional Orthopedics. Interventional Orthopedics Foundation Annual Meeting. Denver, 2015. https://interventionalorthopedics.org/wp-content/uploads/2017/08/AmnioProducts-Poster.pdf
(2) Becktell L, Matuska A, Hon S, Delco M, Cole B, Fortier L. Proteomic analysis and cell viability of nine amnion-derived biologics. Orthopedic Research Society Annual Meeting, New Orleans, 2018. https://app.box.com/s/vcx7uw17gupg9ki06i57lno1tbjmzwaf
(3) Panero, A, Hirahara, A., Andersen, W, Rothenberg J, Fierro, F. Are Amniotic Fluid Products Stem Cell Therapies? A Study of Amniotic Fluid Preparations for Mesenchymal Stem Cells With Bone Marrow Comparison. The American Journal of Sports Medicine, 2019 47(5), 1230–1235. https://doi.org/10.1177/0363546519829034
(4) Soltani SK et al. Safety and efficacy of allogenic placental mesenchymal stem cells for treating knee osteoarthritis: a pilot study. Cytotherapy, Volume 21, Issue 1, 54 – 63. https://www.celltherapyjournal.org/article/S1465-3249(18)30673-X/fulltext
(5) Jin JH, et al. Comparative Analysis of Human Mesenchymal Stem Cells from Bone Marrow, Adipose Tissue, and Umbilical Cord Blood as Sources of Cell Therapy. Int. J. Mol. Sci. 2013, 14(9), 17986-18001; https://doi.org/10.3390/ijms140917986
(6) Wu T, Dong Y, Song Hx, Fu Y, Li JH. Ultrasound-guided versus landmark in knee arthrocentesis: A systematic review. Semin Arthritis Rheum. 2016 Apr;45(5):627-32. doi: 10.1016/j.semarthrit.2015.